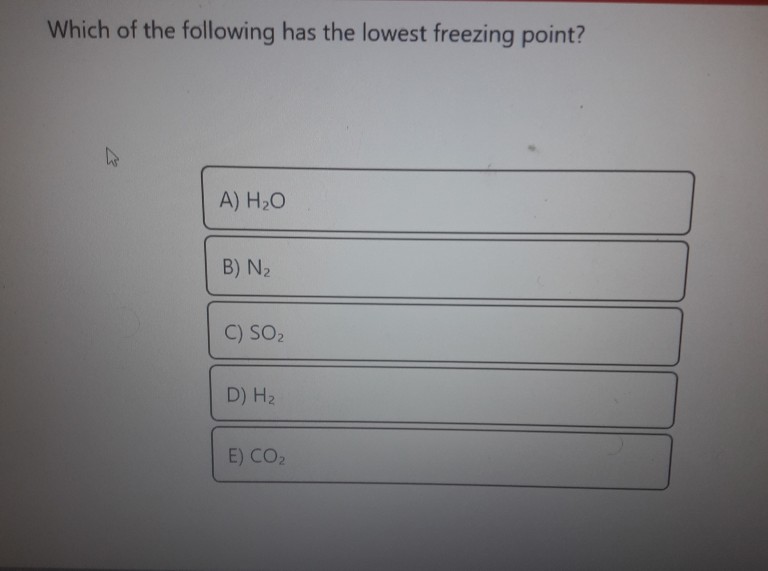
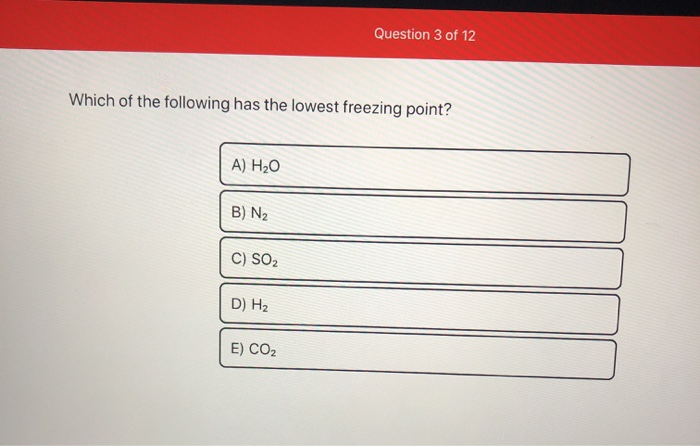
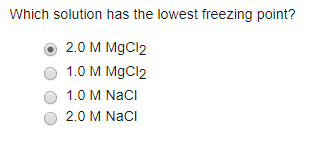SOLVED: Which aqueous solution will have the lowest freezing point? A. 0.1 m MgCl2 B. 0.1 m NaCl C. 0.1 m Na3PO4 D. pure H2O

Which of the following 0 1M aqueos solution will have the lowest freezing point - K2SO4, NaCl,urea and glucose - Chemistry - Solutions - 13781011 | Meritnation.com