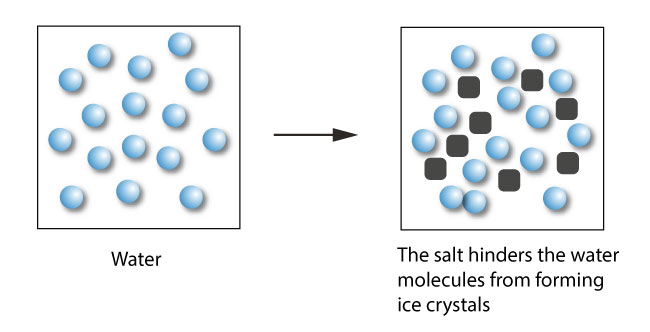
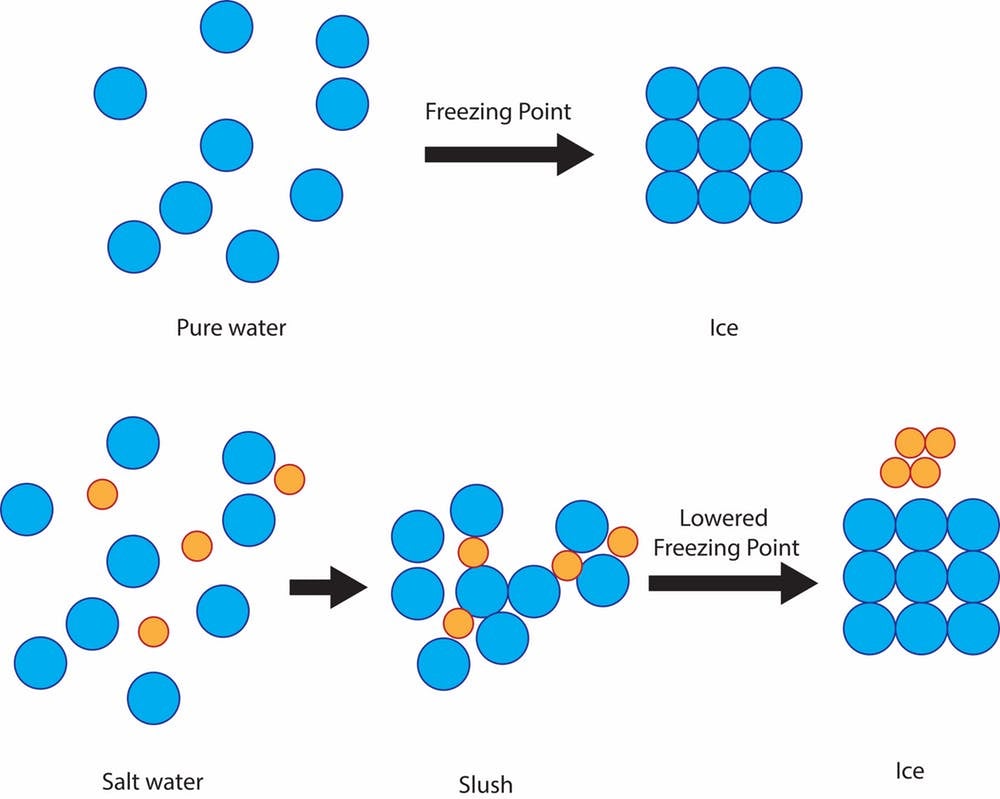
TLDSB Resources on Twitter: "Inter/Sr Interactive Chemistry https://t.co/NY0LnNHI3q Salt Lowers the Freezing Point of Water - Demonstrates how sodium chloride (salt) reduces the freezing point of water.#tldsblearns https://t.co/NtO5GgUP7p" / Twitter

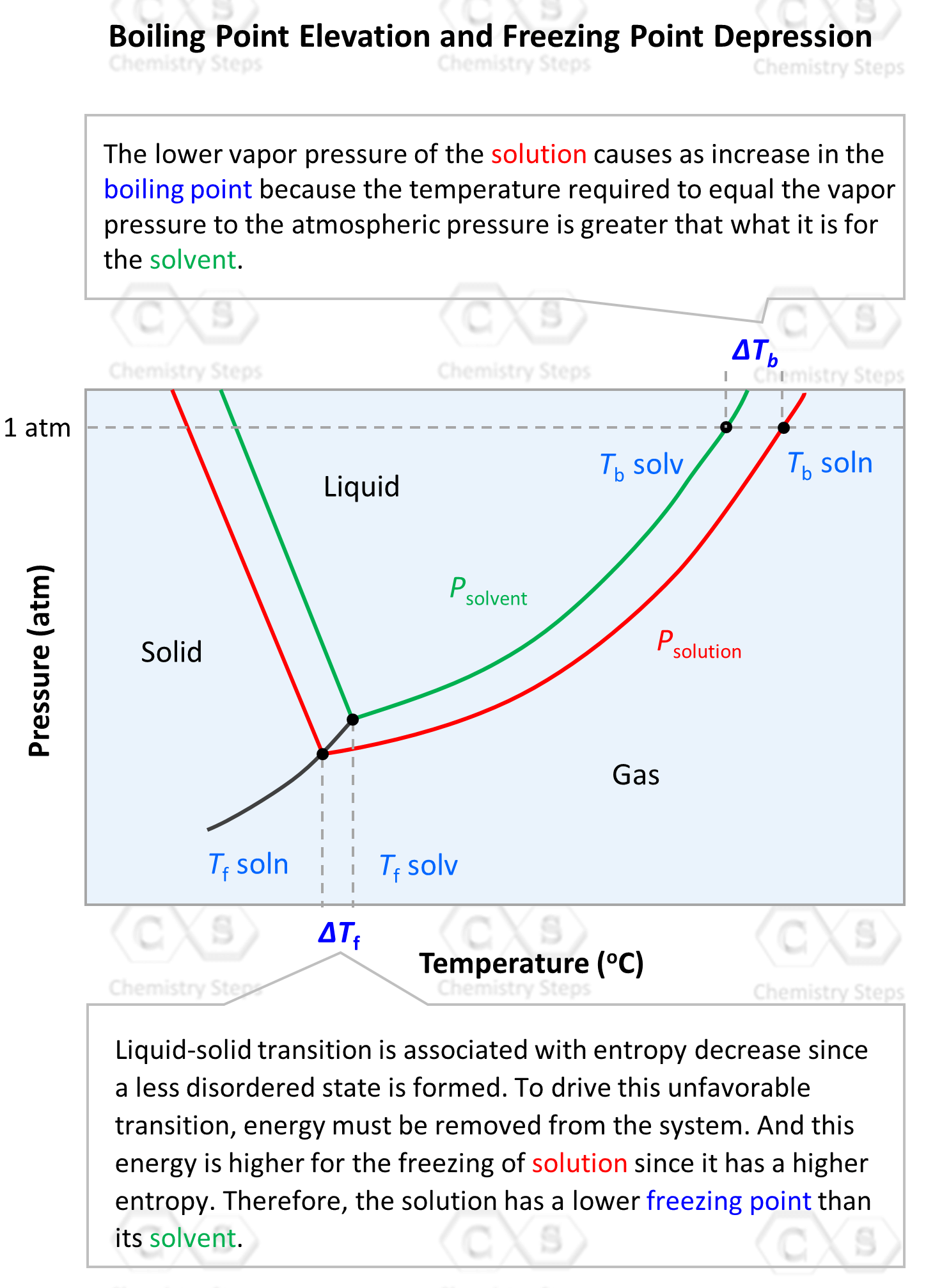
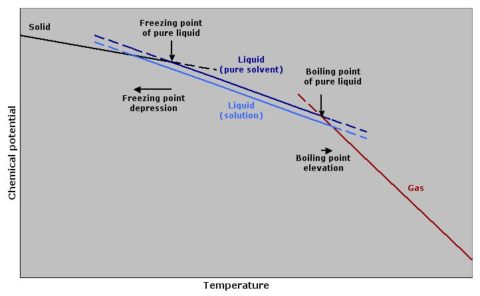
SOLVED: Compared to pure water; 1 M salt-water solution Will have the same boiling point and freezing point B, lower boiling point and lowrer freezing point higher boiling point and lower freezing

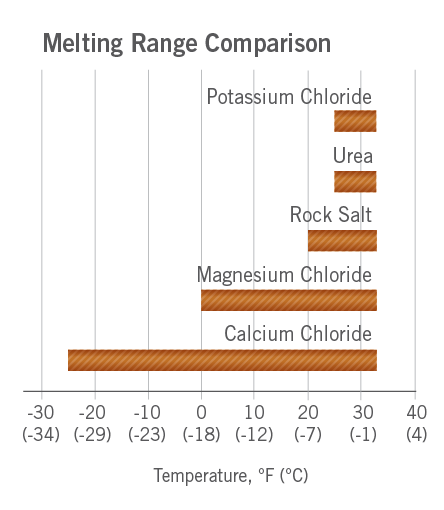

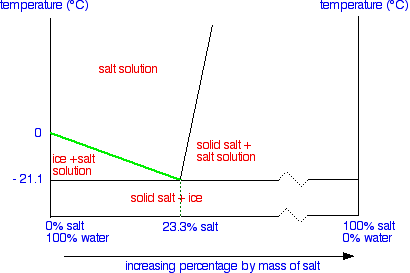


:max_bytes(150000):strip_icc()/two-glasses-full-of-crushed-ice-with-frost-on-outside-of-one--melting-ice-below--and-heap-of-salt-98358220-56f9642c3df78c78419330a7.jpg)