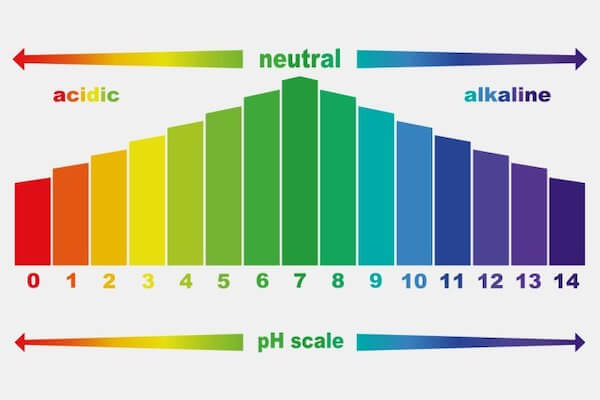
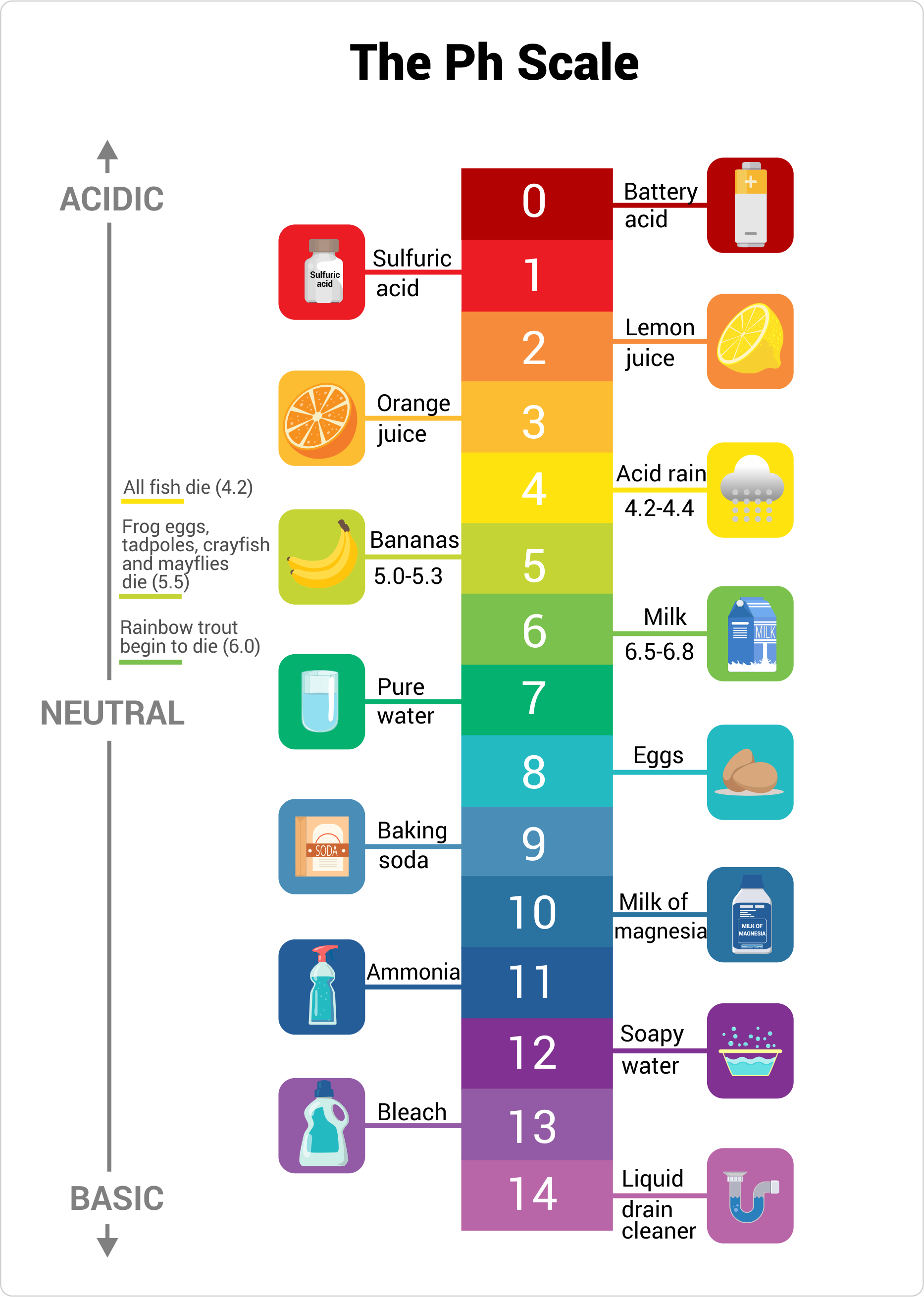
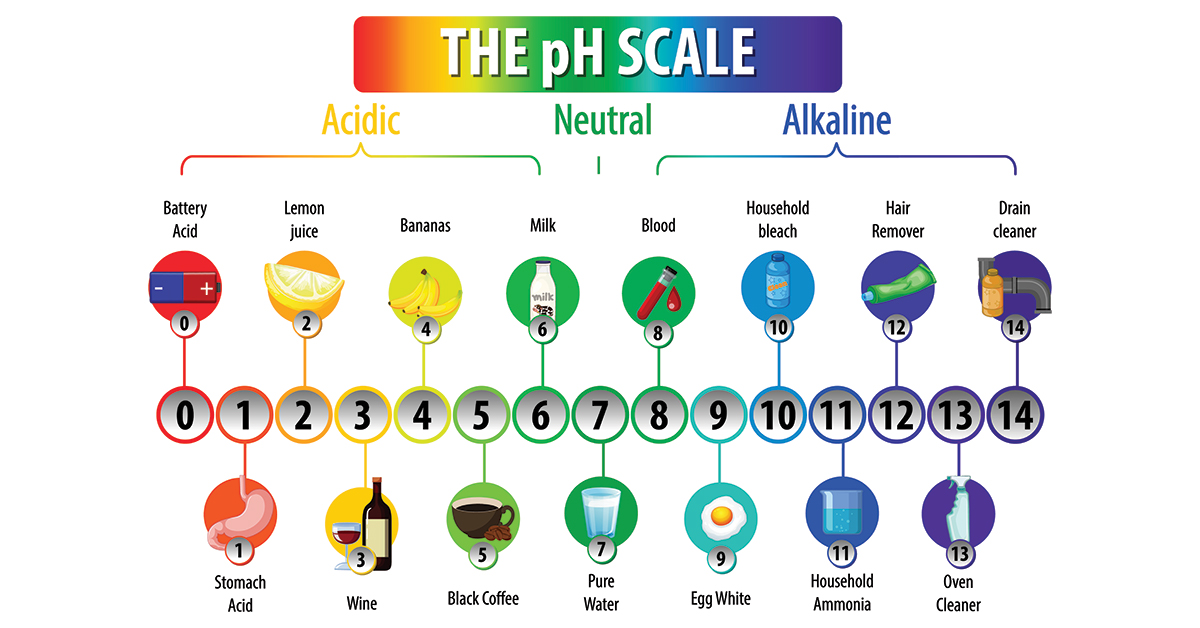
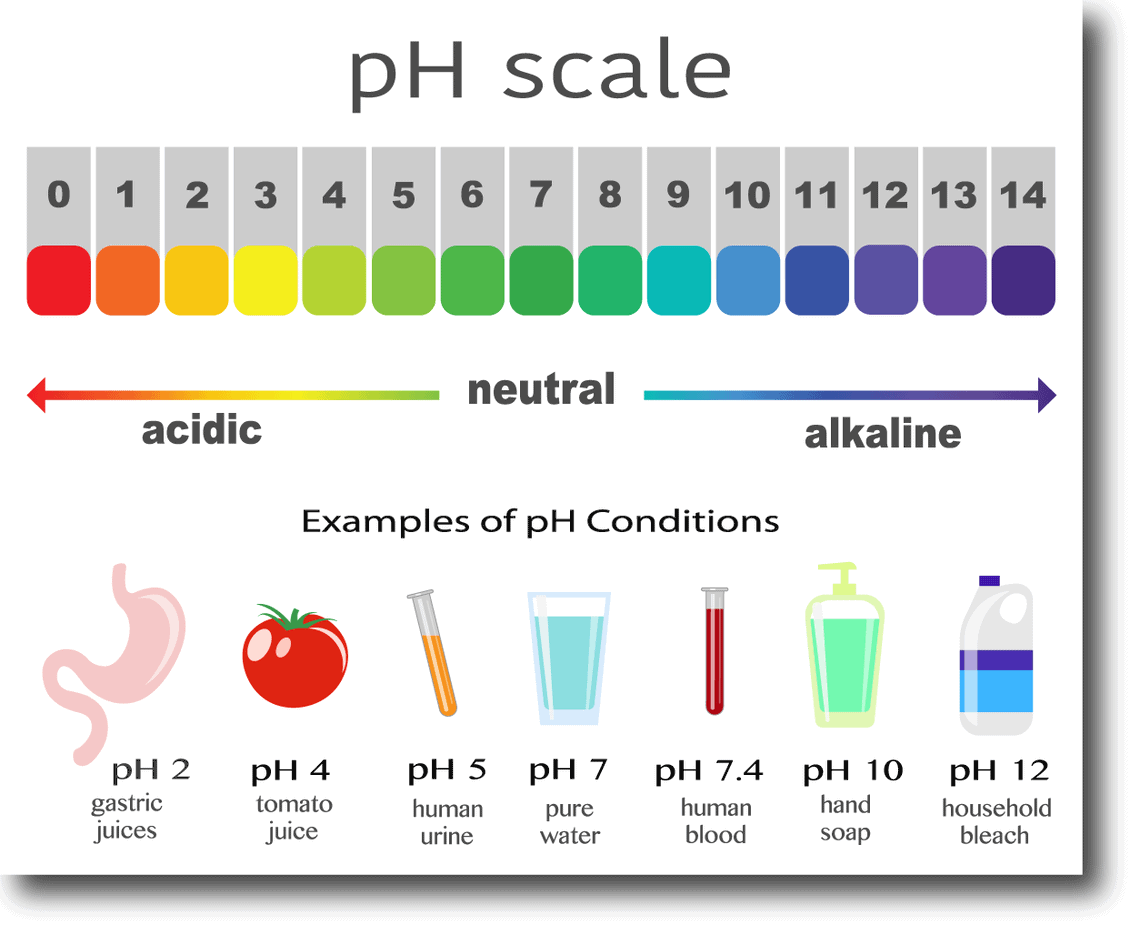
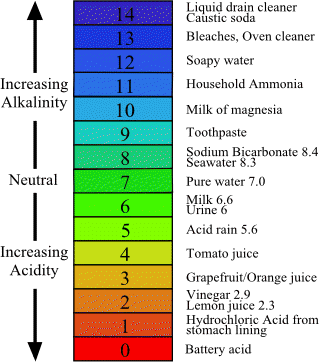
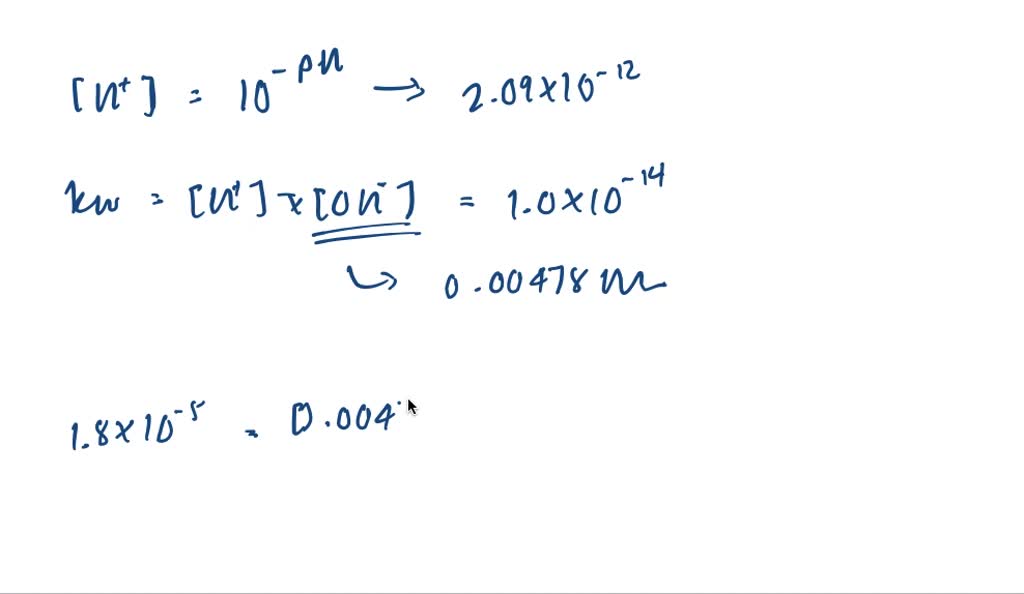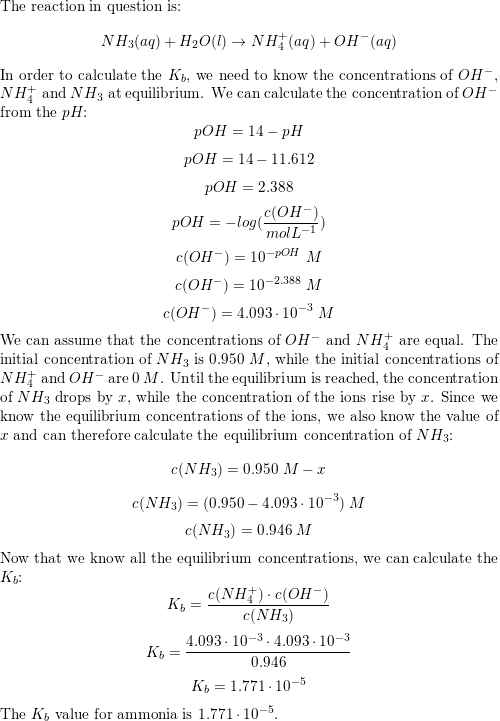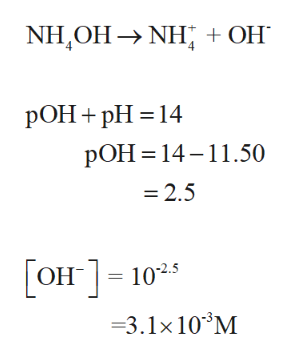![SOLVED: 15. The pH of household ammonia is 11.9. The pH of a chemical solution is given by Ihe formula pH = logio[H+] where [H:] is the concentralion of hydrogen ions, What SOLVED: 15. The pH of household ammonia is 11.9. The pH of a chemical solution is given by Ihe formula pH = logio[H+] where [H:] is the concentralion of hydrogen ions, What](https://cdn.numerade.com/ask_previews/eeece973-1e1f-4052-97f6-4fd76b3feaa6_large.jpg)
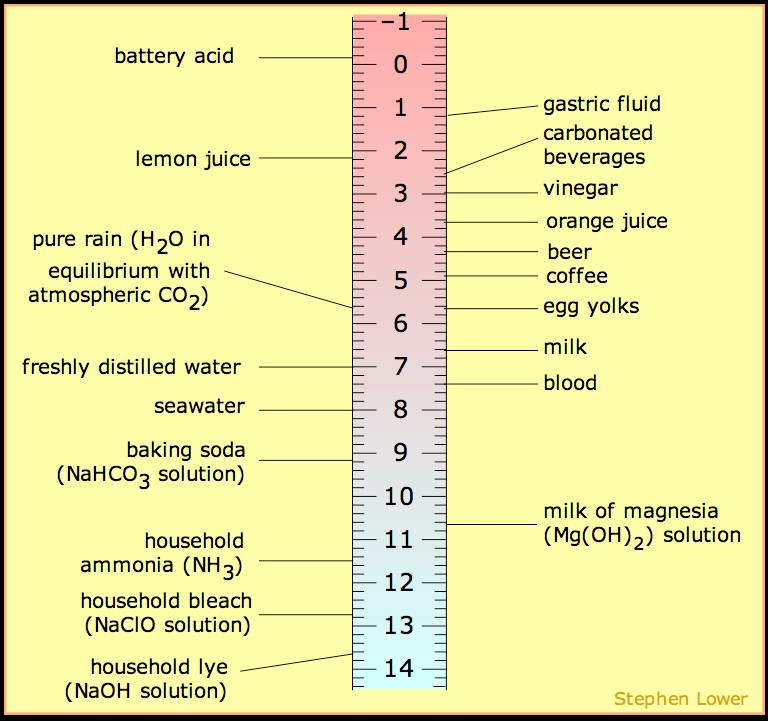
SOLVED: 15. The pH of household ammonia is 11.9. The pH of a chemical solution is given by Ihe formula pH = logio[H+] where [H:] is the concentralion of hydrogen ions, What

SOLVED:The pH of a solution of household ammonia, a 0.950 M solution of NH3, is 11.612 . Determine Kb for NH3 from these data.