
Global Sterilization Standards Support Infection Prevention Better Patient Outcomes - Transforming Outcomes

ASTM E1837-96(2014) - Standard Test Method to Determine Efficacy of Disinfection Processes for Reusable Medical Devices (Simulated Use Test)

Sustainability | Free Full-Text | Green Servitization in the Single-Use Medical Device Industry: How Device OEMs Create Supply Chain Circularity through Reprocessing

New Requirements for Medical Device Cleaning Validations: An Overview of ST98 - Medical Design Briefs

ASTM E1837-96 - Standard Test Method to Determine Efficacy of Disinfection Processes for Reusable Medical Devices (Simulated Use Test)



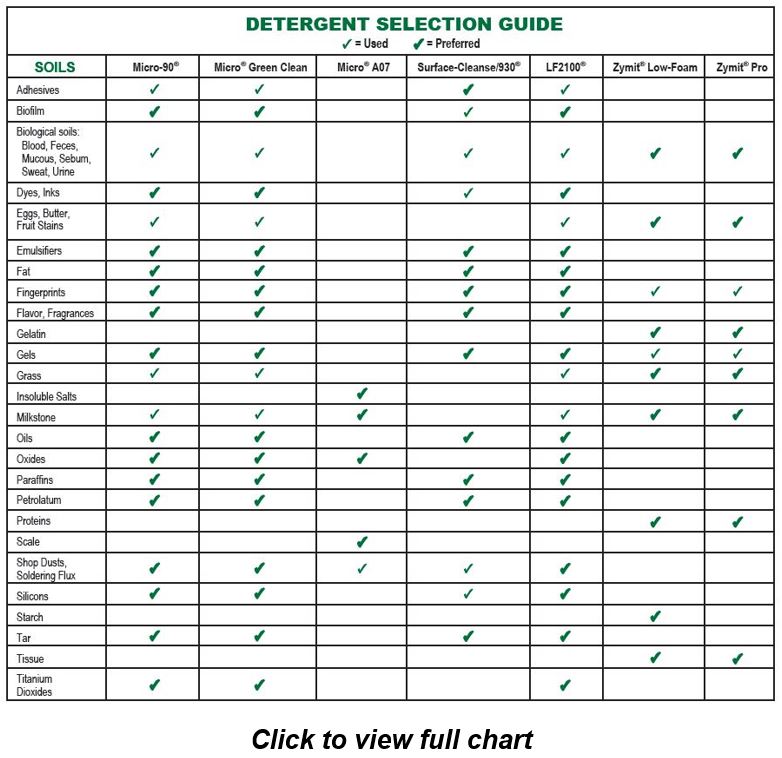


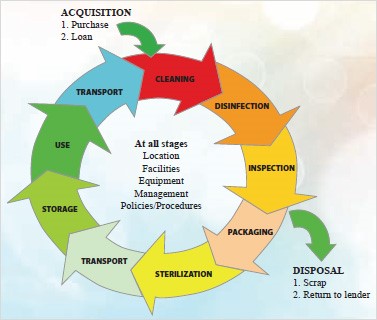









![Medical device reprocessing cycle for a critical medical device [4] | Download Scientific Diagram Medical device reprocessing cycle for a critical medical device [4] | Download Scientific Diagram](https://www.researchgate.net/publication/344536728/figure/fig1/AS:962839208554503@1606570164411/Medical-device-reprocessing-cycle-for-a-critical-medical-device-4.png)
