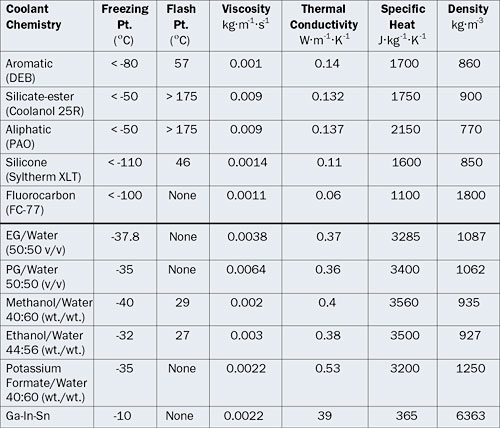
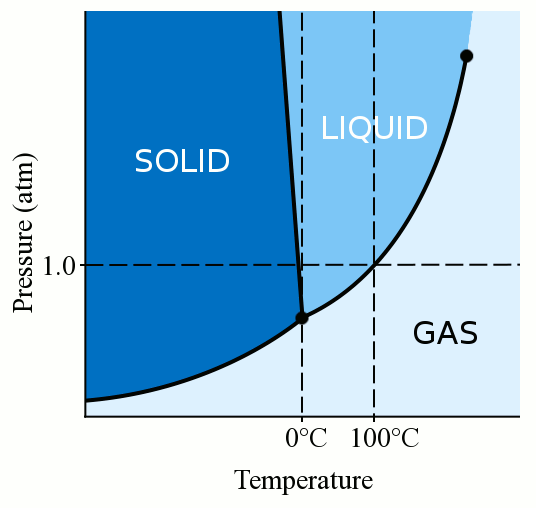
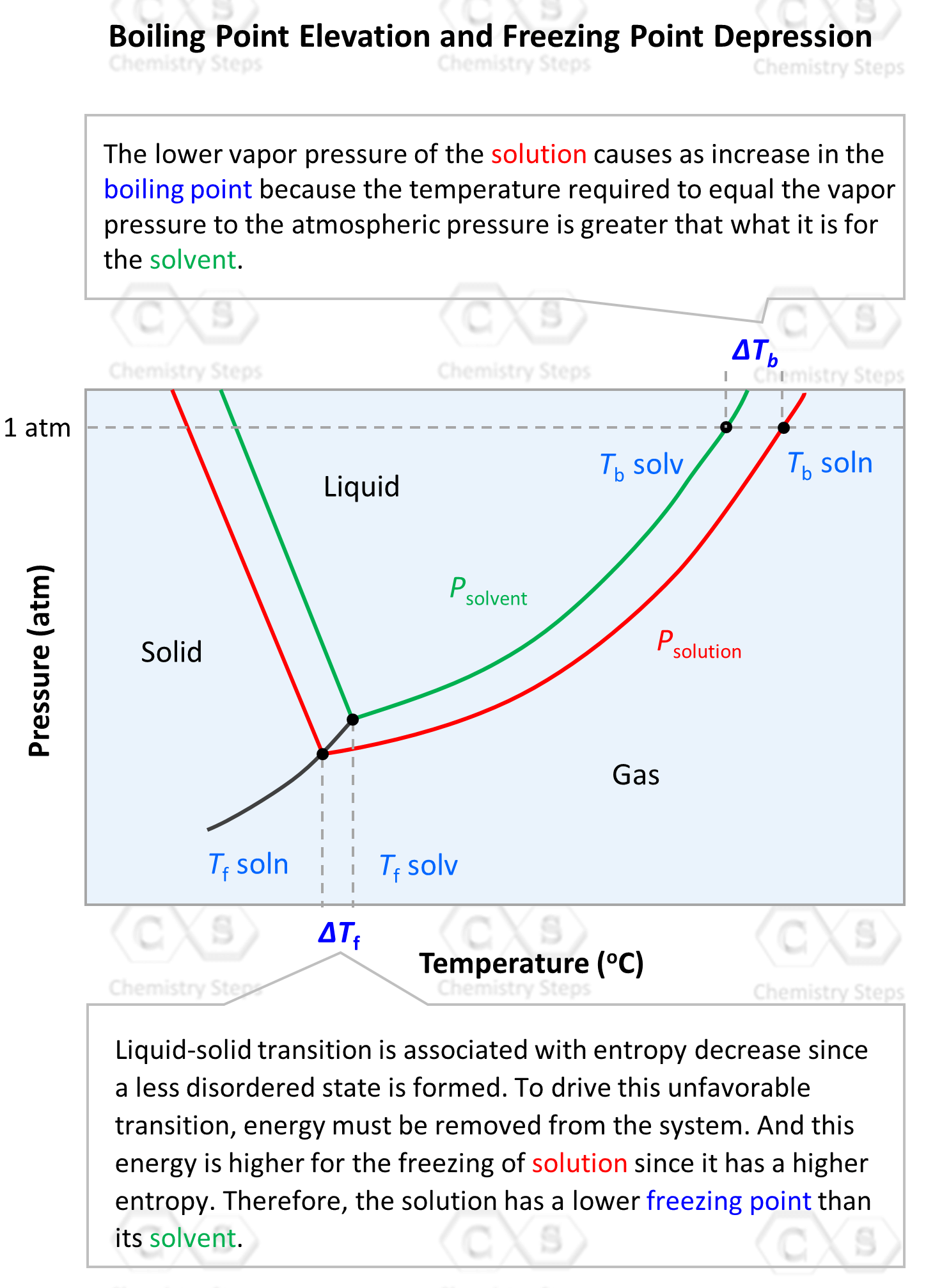
Freezing Point Determination of Water–Ionic Liquid Mixtures | Journal of Chemical & Engineering Data
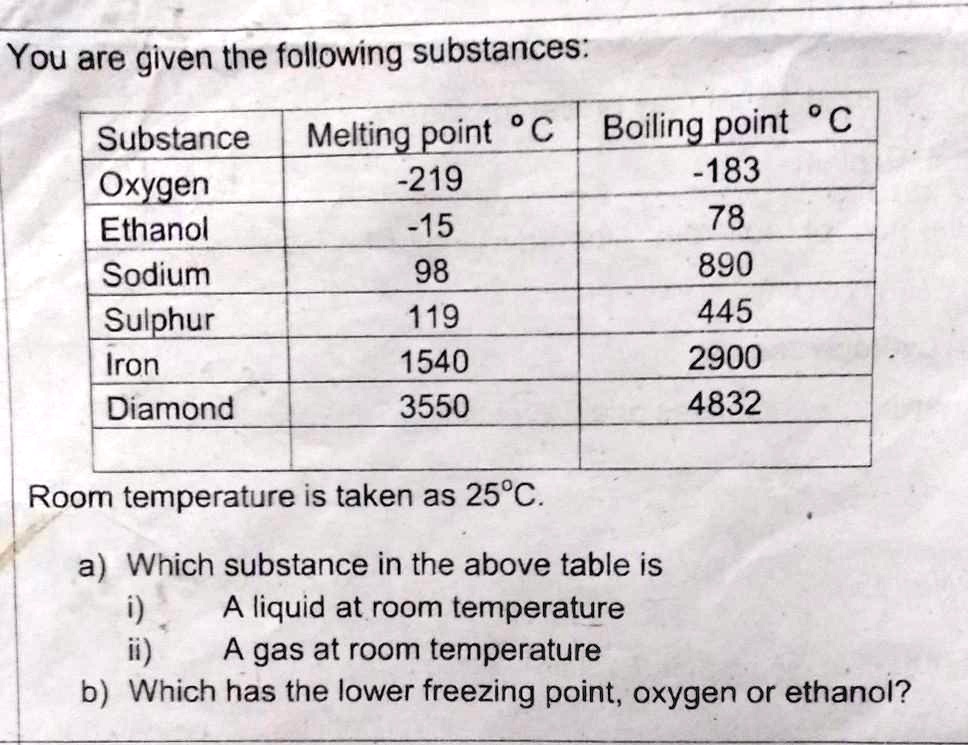
SOLVED: 'give the answer of of this question please You are given the following substances: point C Boiling point C Substance Melting -219 -183 Oxygen Ethanol -15 78 Sodium 98 890 Sulphur

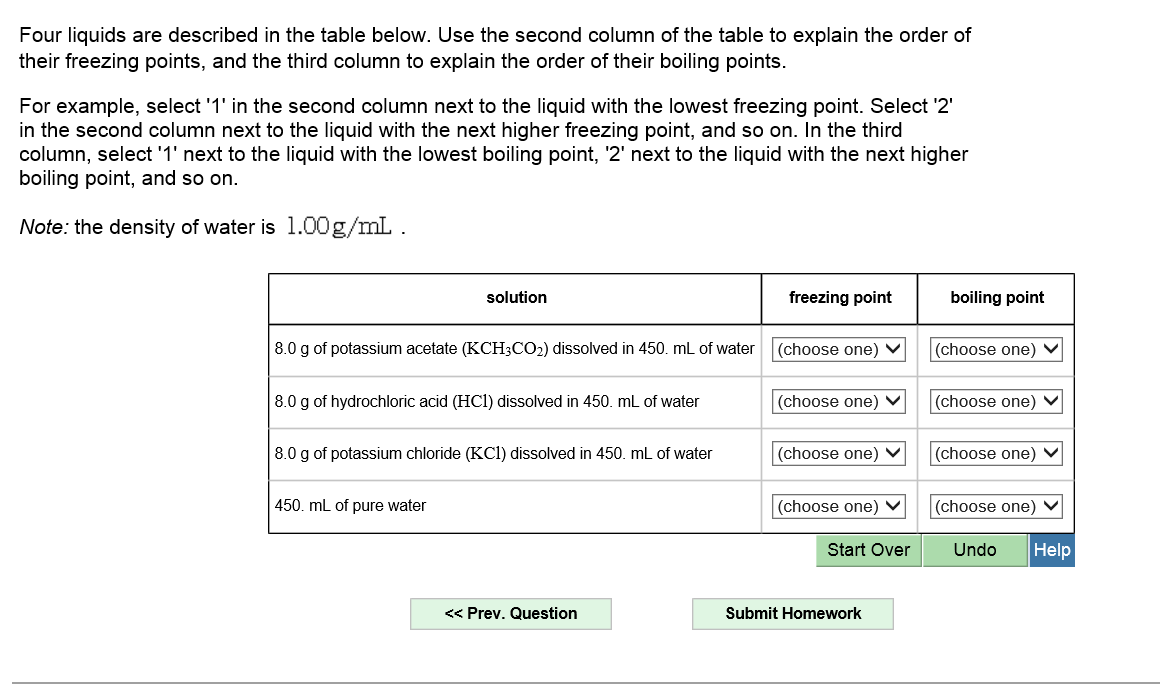
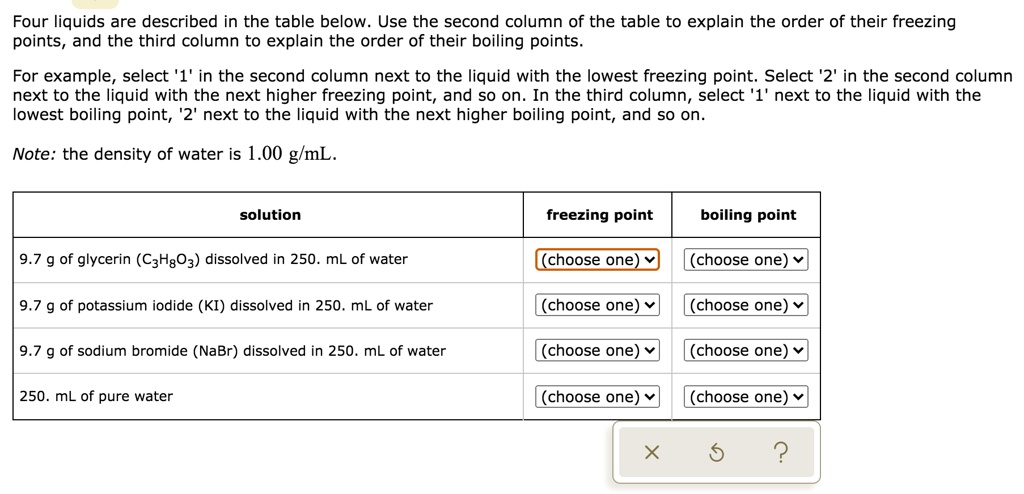
SOLVED: Four liquids are described in the table below. Use the second column of the table to explain the order of their freezing points and the third column to explain the order
its normal boiling point is -189°C. Oxygen is a gas at room temperature. If the normal melting point of a substance is below room temperature, the substance is a liquid at room temperature. Benzene melts at 6°C and boils at 80°C; it is a liquid at room ...
13.9: Freezing Point Depression and Boiling Point Elevation- Making Water Freeze Colder and Boil Hotter - Chemistry LibreTexts
:max_bytes(150000):strip_icc()/the-freezing-point-of-water-609418_FINAL-01f50f5f4f7d4a39854bebcc59df1aa4.gif)