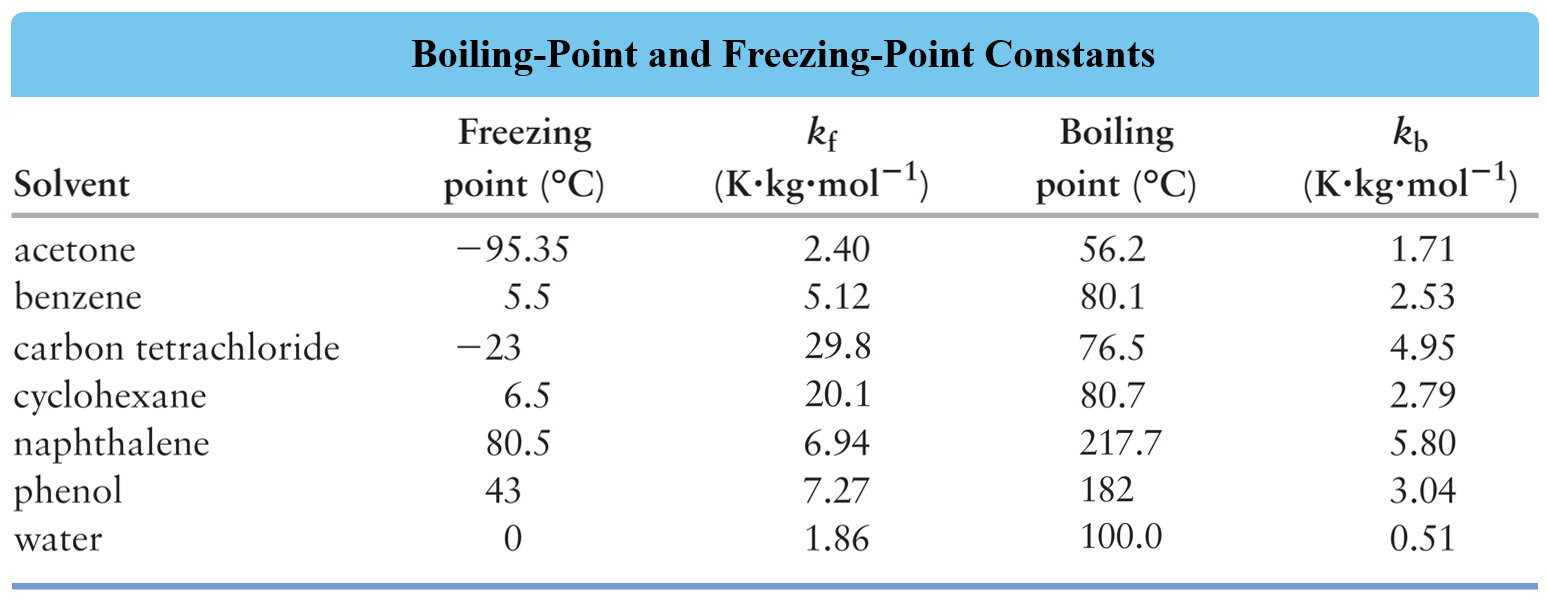
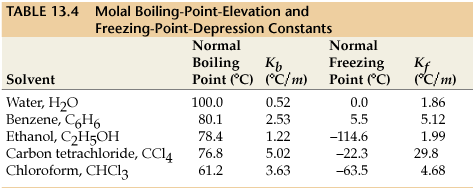
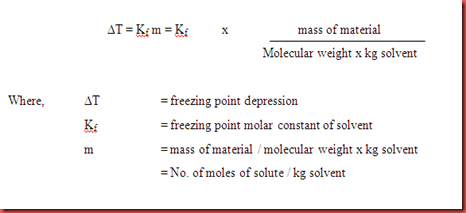Calculate the freezing point depression and boiling point elevation of a solution of 10.0 g of urea (M(B)=60) in 50.0 g of water at 1 atm. pressure.K(b) and K(f) for water 0.52^(@)C

How do you find the freezing point of pure water from the freezing point depression equation? | Homework.Study.com

Difference Between Freezing Point Depression and Boiling Point Elevation | Compare the Difference Between Similar Terms

Determine the freezing point depression of H_2O in 1.50 M solution of C_{12}H_{22}O_{11}? | Homework.Study.com