Experimental study on the competition between carbon dioxide hydrate and ice below the freezing point - ScienceDirect
![SOLVED: When 1.00 g of urea [CO(NH2)2] is dissolved in 200 g of solvent A, the A freezing point is lowered by 0.250*C. When 1.50 g of the nonelectrolyte Y is dissolved SOLVED: When 1.00 g of urea [CO(NH2)2] is dissolved in 200 g of solvent A, the A freezing point is lowered by 0.250*C. When 1.50 g of the nonelectrolyte Y is dissolved](https://cdn.numerade.com/ask_previews/8c9cb3ee-04e1-472c-acbd-e31522af0181_large.jpg)
SOLVED: When 1.00 g of urea [CO(NH2)2] is dissolved in 200 g of solvent A, the A freezing point is lowered by 0.250*C. When 1.50 g of the nonelectrolyte Y is dissolved

Antifreeze Refractometer - 3-In-1 Coolant Tester for Checking Freezing Point, Co 744110055290 | eBay
The phase rule and its applications . c point at whichthe inactive mixture consisting of equal amounts of d- and1- form melts. Owing to the similar effect of the one form onthe

ASTM D1177 Automatic Freezing Point Tester for Engine Coolant - China Freezing Point Tester and Automatic Freezing Point Tester

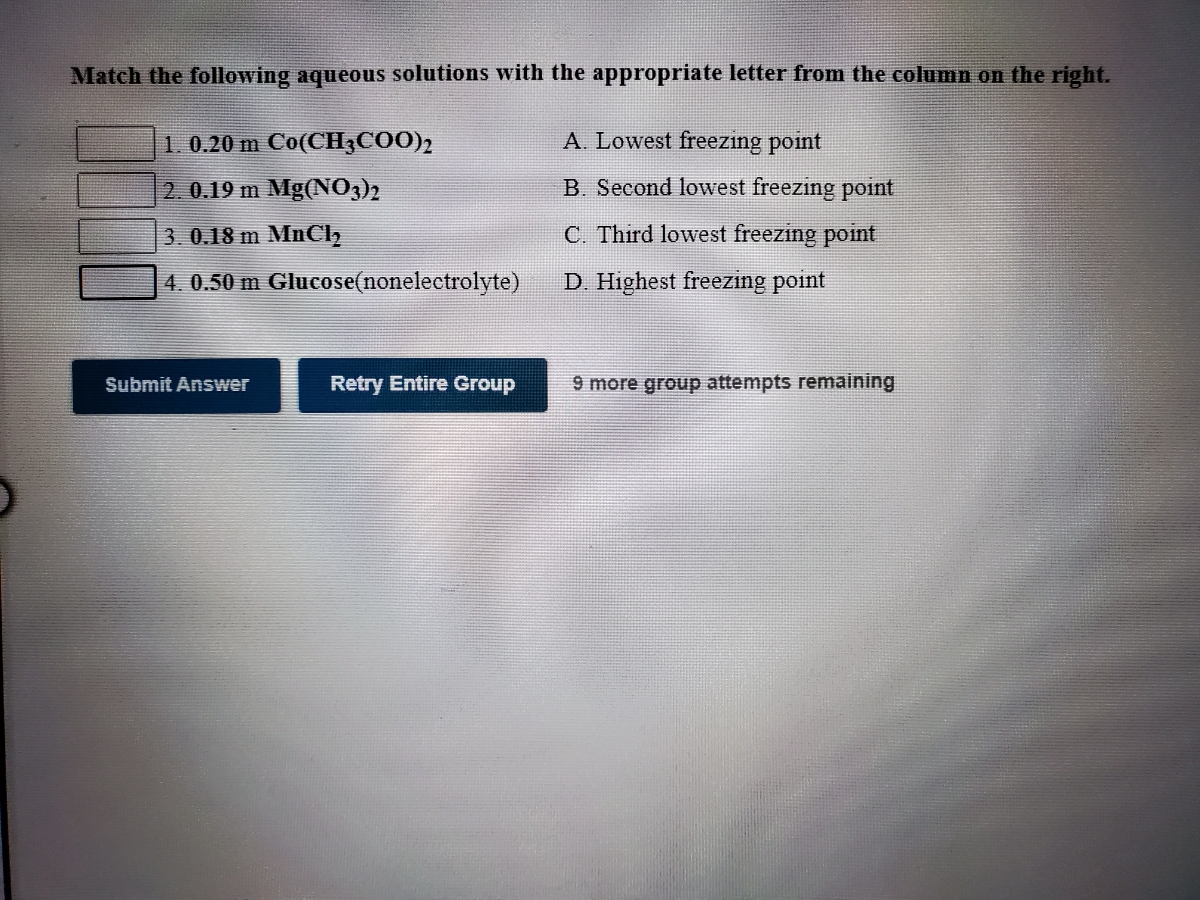
SOLVED:Predict the approximate freezing points of 0.10 m solutions of the following solutes dissolved in water: (a) CO(NH2)2( urea ) ; (b) NH4 NO3 ; (c) HCl ; (d) CaCl2 (e) MgSO4 ; (

The Freezing-point, Boiling-point and Conductivity Methods. By Harry C. Jones, Instructor in Physical Chemistry in Johns Hopkins University. Easton, Pa., Chemical Publishing Co. Pp. 64. Price, 75 cents. | Science

.webp)