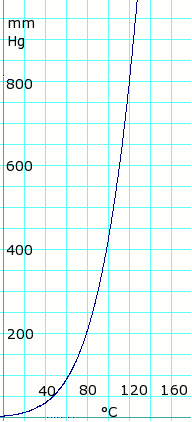
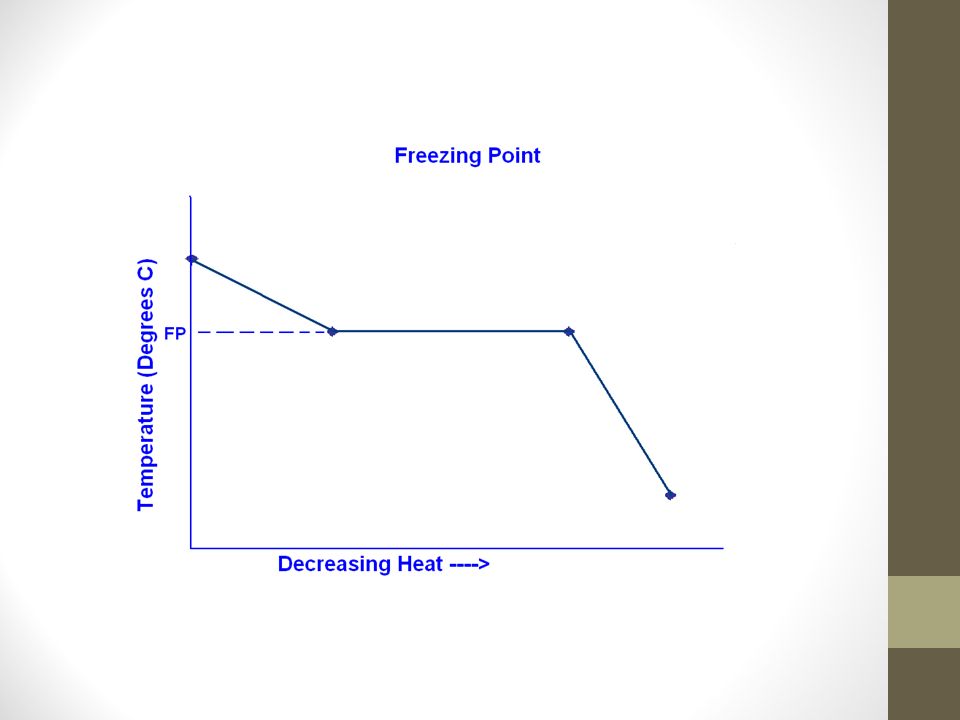
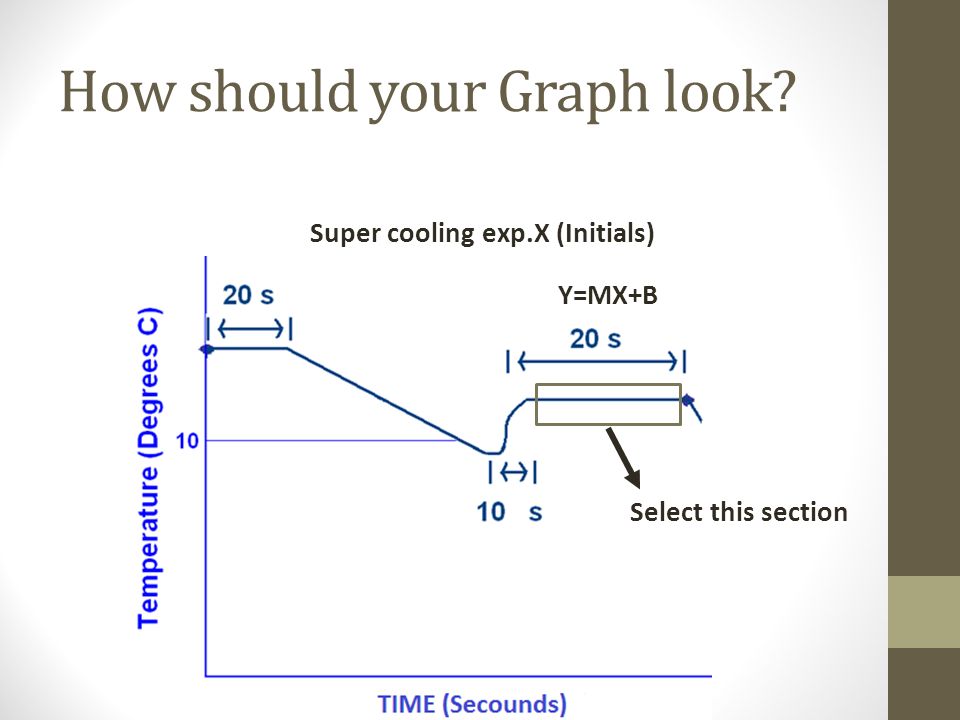
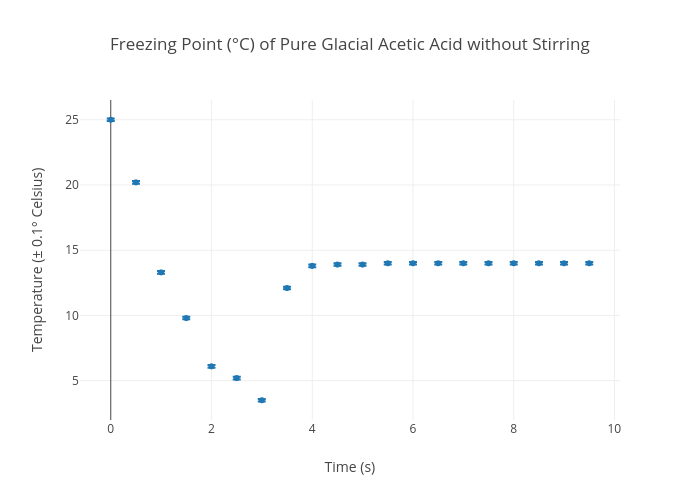
The freezing point of solution containing 0.2 g of acetic acid in 20.0 g of benzene is lowered by 0.45^(@)C. Calculate the degree of association of acetic acid in benzene. (K(f)=5.12 K^(@)

The depression in freezing point of water observed for the same amount of acetic acid, trichloroacetic acid and trifluoroacetic acid increases in the order given above. Explain briefly.

The depression in freezing point of water observed for the same amount of acetic acid, trichloroacetic acid and trifluoroacetic acid increases in the order given above. Explain briefly.

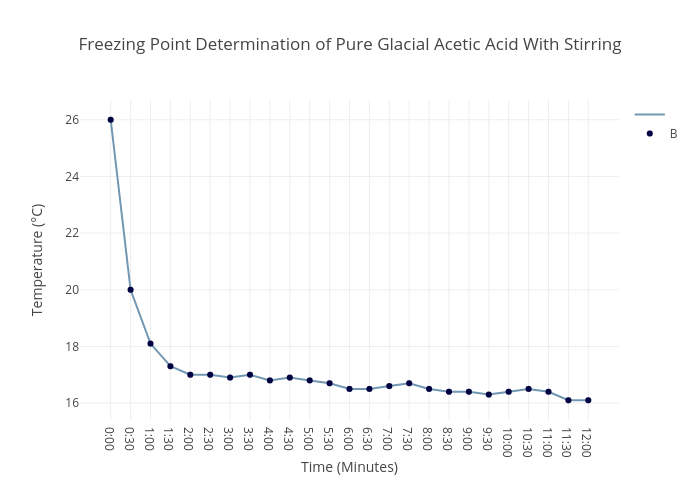
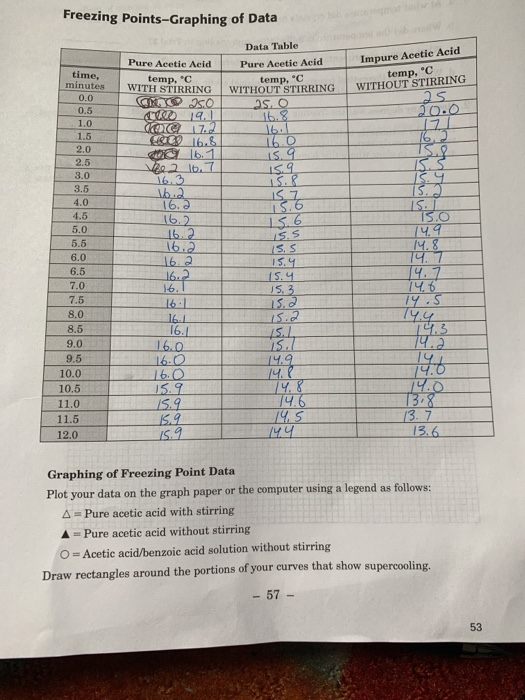
Solved) - graph the data by hand and answer the following questions: 1. a)... (1 Answer) | Transtutors
What happens to the freezing point and boiling point of acetic acid and water when solute is added? - Quora

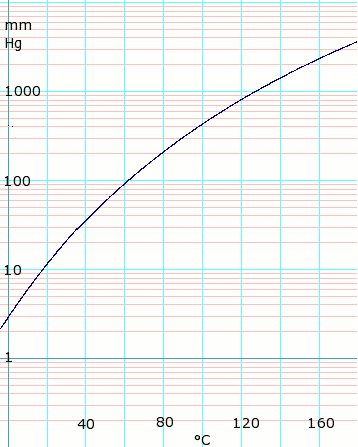
Determination of the molar mass of acetic acid in benzene using freezing point depression is - YouTube
The freezing point of a solution containing 0.2g of acetic acid in 20.0 g benzene is lowered by 0.45ºC. - Sarthaks eConnect | Largest Online Education Community
How could the fact that vinegar evaporates without leaving concentrated acetic acid behind, be explained to a non-chemist? - Quora